20. February 2023 : Clinical trials 23 : results to look forward!

20.02.2023 : The most interesting clinical trials coming to an end in 2023… Recently, Nature Medicine reporters C. Arnold and P. Webster asked leading researchers to name the most interesting clinical trials coming to an end in 2023. Roger Albin, Professor of Neurology and co-director of the Movement Disorders Clinic at the University of Michigan Medical School, highlighted the Exenatide phase […]
31. January 2023 : CTIS : a single-entry point for EU clinical trials

31.01.2023 : CTIS : a single-entry point for EU clinical trials Today, January 31st 2023, the Clinical Trial Information System (CTIS) becomes mandatory as the single-entry point for the submission and assessment of clinical trial data for sponsors and regulators in the European Union (EU). The new Clinical Trial Regulation (CTR) (EU) No 536/2014 entered into application a year ago […]
Enriched stable 204Pb as tracer at ultra-low levels in clinical investigations
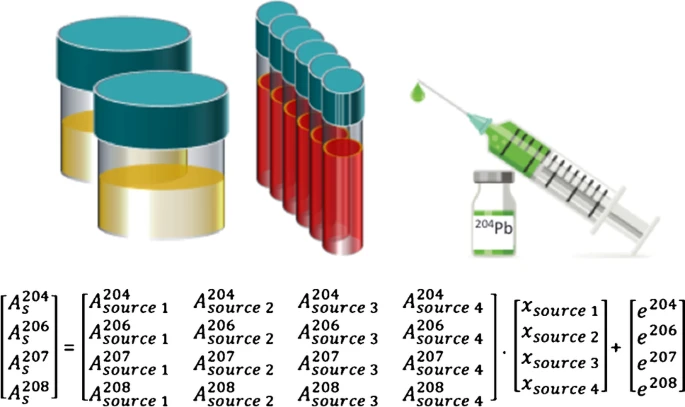
Abstract The potential of enriched Pb (204Pb) was assessed to monitor pathways of trace levels of Pb in the pg range within the human body via isotope pattern variation in situations where natural lead cannot be used as a tracer due to regulatory limitations. Isotope ratio measurements were accomplished by means of (multi-collector) inductively coupled […]
04. April 2022 : The HEART Approach for Clinical Trial Leadership

PRESS RELEASE Project management vs Project leadership New drug development remains highly risky and costly despite the fact that approval success rates increased to 62% in 2017 (Galson et al., Nature, 2021). Galson et al. name poor execution and inadequate project management as one of the factors causing every third study to fail in phase […]
1. April 2022 : Neuigkeiten am Corona Horizont

💡Neuigkeiten am Corona Horizont: 🦠 Das Paul-Ehrlich-Institut hat in seinem aktuellen Newsletter 03/2022 von 1 April 2022 folgende Neuigkeiten zu Neuzulassungen in der Covid19 Prophylaxe und Impfstoffe bekanntgegeben: Antrag auf bedingte Zulassung für den proteinbasierten COVID-19-Impfstoffkandidaten Vidprevtyn gestellt👉https://lnkd.in/e3KQJqdE Rolling-Review für Booster-Impfstoff COVID-19 Vaccine HIPRA hat begonnen👉https://lnkd.in/eChgTDbd Zulassung der monoklonalen Antikörper-Kombination Evusheld (Tixagevimab und Cilgavimab) zur COVID-19-Prophylaxe erteilt👉https://lnkd.in/ef9aCnM2
8. November 2021: Advancing Research : Gouya Insights holds Free Expert Consultation for clinical trials

PRESS RELEASE Advancing Research: Gouya Insights holds Free Expert Consultation for clinical trials Vienna, 08.11.2021 – Clinical trials are a central component of medical research. Without such trials, there would be no approved drugs and medical development would come to a standstill. PD Dr. Ghazaleh Gouya Lechner wants to further promote the development of clinical […]
Daten sind Treibstoff für Forschung

Registerdaten können einen entscheidenden Beitrag zu Innovation und Stärkung des Forschungsstandortes Österreich leisten. Drei Expert*innen erklären ihren Standpunkt. ( Priv. Doz. Dr. Johaness Pleiner-Duxneuner / Ghazaleh Gouya-Lechner / Michael Strassnig )
PHARMAFORSCHUNG – Die Presse. Daten sind Treibstoff für Forschung

Pharmastandort. Registerdaten können einen entscheidenden Beitrag zu Innovation und Stärkung des Forschungsstandortes Österreich leisten. Drei Expert*innen erklären ihren Standpunkt. (Priv. Doz. Dr. Johaness Pleiner-Duxneuner / Ghazaleh Gouya-Lechner / Michael Strassnig) Similar article : https://gouya-insights.com/daten-sind-treibstoff-fur-forschung/
6. September 2021: Medical and Scientific Writing

Medical and Scientific Writing Information hub for the pharmaceutical industry The development of drugs or medical devices to market approval is a years-long development process with extensive regulatory requirements to prove efficacy and safety. Special experts, called medical writers, prepare the results and data for the regulatory authorities. Dr. Joana Enes from Gouya Insights explains what […]
Remote-Arbeiten in der Forschung

PHARMAustria sprach mit Priv.-Doz. Dr. Ghazaleh Gouya Lechner, Vorstandsmitglied der Österreichischen Gesellschaft für Pharmazeutische Medizin (GPMed), über die Situation der medizinischen Forschung in Pandemiezeiten, das vermehrte Remote-Arbeiten sowie die zukünftige Rolle von Artificial Intelligence bei klinischen Studien. (s.44)

